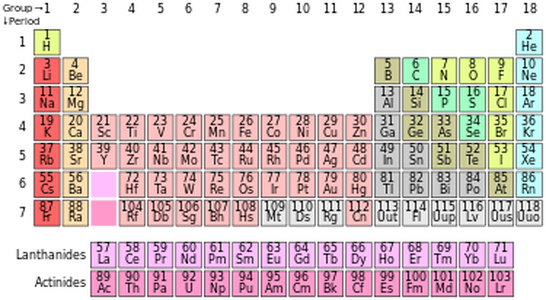
Here is a standard Periodic table where the elements are arranged according to their atomic numbers. Columns are called groups and rows are called periods. Excluding the transition metals, elements in the same group have the same number of valence electrons and as you go across a period you are adding another valence electron. Elements in the same period have electrons in the same number of electron shells, and this number is the period number. For example, all the elements in period three have electrons in three electron shells. This also means that as you go down a group you are adding a new electron shell. Since there are trends in the electron configurations of the elements arranged in the Periodic table, there are also going to be trends with the chemical properties of these elements because these properties are dependent on the activity of the electrons within the atoms
