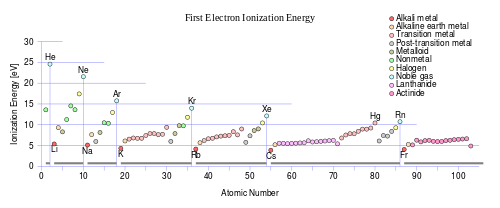
First ionisation energy is the energy required to remove the highest energy electron from an atom. As shown in figure 1 above, the energy required to remove an electron from an atom increases as you go from metals to non-metals. This is because core charge also increases in the same direction, therefore the valence electrons are held in much tighter by the attractive forces for the non-metals then for the metals. Consequently, you would need more energy to remove the highest energy electron from a non-metal than a metal. The figure above also shows that first ionisation energy decreases as you go down a group. Remembering from the atomic radius explanation, as you go down a group you are adding more electron shells and therefore the valence electrons are affected by shielding, thus reducing the actual effective core charge. Therefore, as you go down a group, the core charge stays the same but the valence electrons become less affected by it. So it would be easier to remove an electron from one of the bottom elements of a group then from the top because the valence electrons are not affected as much by core charge.