|
Electronegativity is the ability of an atom to attract electrons to itself.
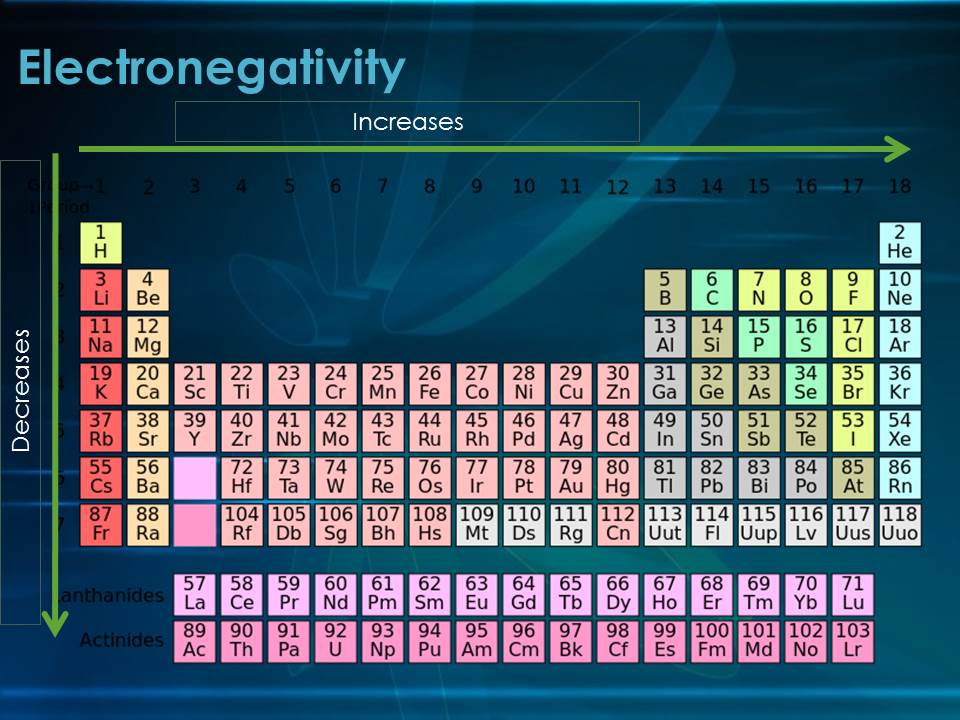
As shown in figure 1 as you go down a group electronegativity decreases. This is due to the shielding effect(as discussed in previous trends) which decreases the effective core charge as you go down a group. Since the effective core charge is weak the atom will not be very good at attracting other electrons to itself. Therefore electrongeativity will decrease down a group. Also shown is that it increases as you go across a period. Since core charge increases across a period the pull on the outer electrons will increase as you go across a period. Therefore electronegativity will also increase across a period. |