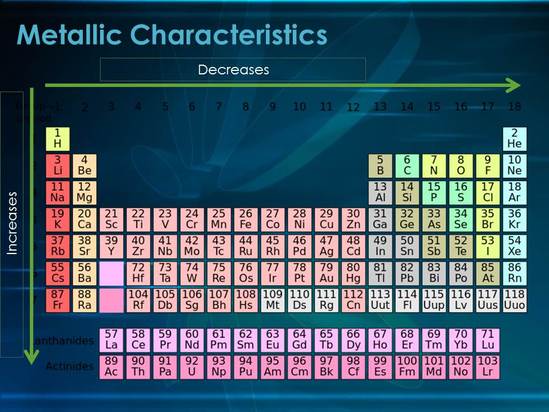
 Figure 1: Metallic characteristics decrease across a period and increase down a group
Figure 1: Metallic characteristics decrease across a period and increase down a group
Metallic characteristics are chemical properties that metallic elements have which are a result from how readily an atom loses electrons.
As shown in Figure 1 metallic characteristics decrease as you go from metals to non-metals as the metals core charge is relatively weak and therefore the electrons are not held in as tightly as in the non-metals.
Metallic Characteristics also increase as you go down a group because the electrons are less tightly held in due to shielding and are therefore easier to lose.
As shown in Figure 1 metallic characteristics decrease as you go from metals to non-metals as the metals core charge is relatively weak and therefore the electrons are not held in as tightly as in the non-metals.
Metallic Characteristics also increase as you go down a group because the electrons are less tightly held in due to shielding and are therefore easier to lose.
