|
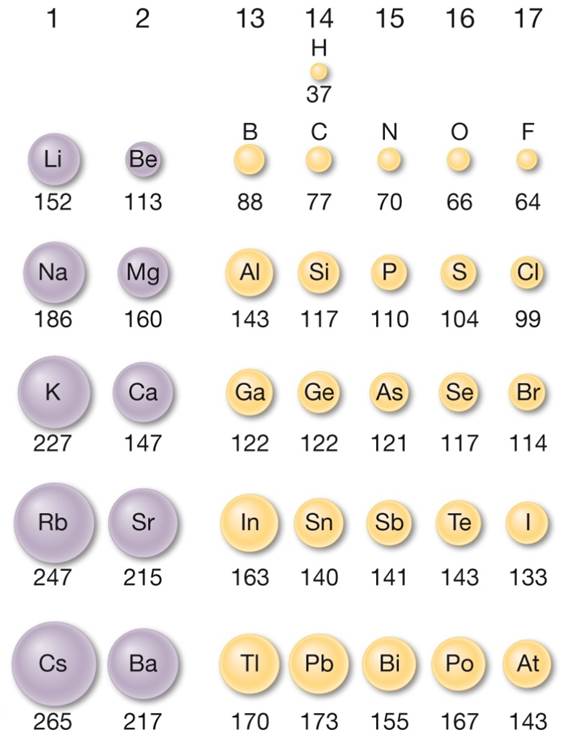
Atomic Radius is the measure of the size of an atom. As you go across a period Atomic Radius decreases and as you go down a Group it increases as shown in Figure 1. This trend can be explained through core charge. As you go across a period the core charge increases, therefore the valence electrons are being pulled in closer to the nucleus making the size of the atom smaller. As you go down a group you add a new electron shell, this means that the valence electrons are further away from the nucleus with more inner shell electrons between them and the nucleus. All these extra electrons reduce the effect of the core charge due to their electron-electron repulsion; this is known as the shielding effect. Due to the shielding effect, the effective core charge on the electron becomes weaker, therefore the force pulling the valence electrons in are weaker than the ones in the previous periods. Thus, the atomic size would be bigger as the electrons are less tightly held in. Figure 2 below shows the relative size of atoms.
|